FDA advises new fees for FY 2022
- 4 August 2021
- Posted by: inetika
- Category: GLOBAL NEWS
No Comments
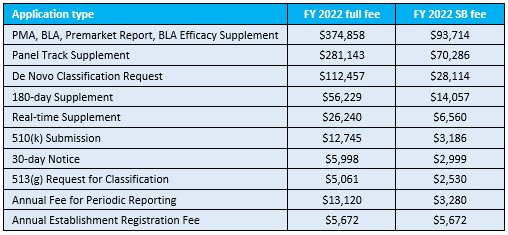
The US Food and Drug Administration has now published its revised user fees for its fiscal year (FY) 2022, which starts on 1 October 2021.
Any submissions or applications arriving at the Agency on or after 1 October 2021 will need to have paid the new fees, which are: